Baby grooming kit OEM: When ‘all-in-one’ means compromised bristle stiffness and hygiene gaps
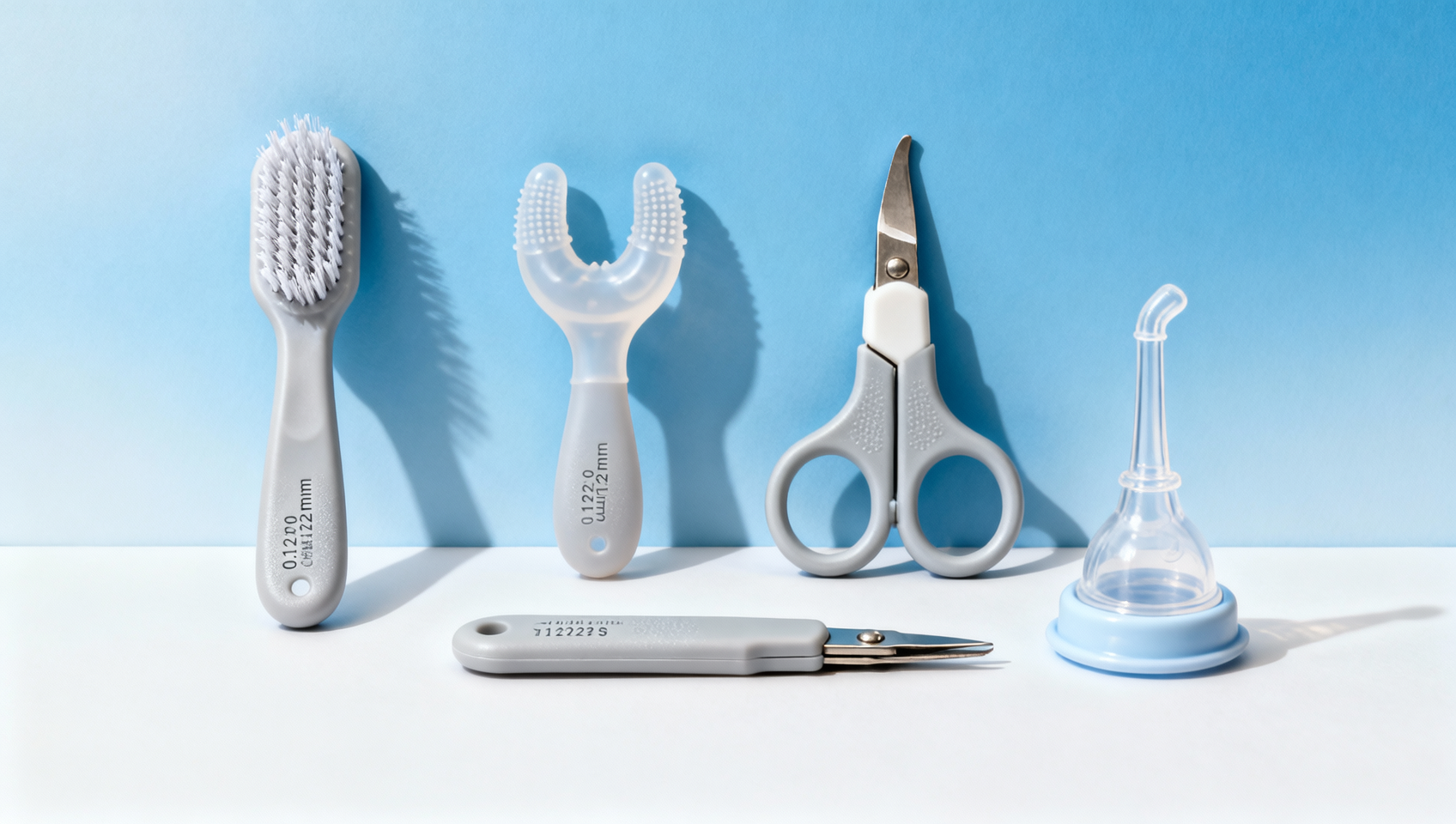
When sourcing a baby grooming kit OEM, 'all-in-one' convenience shouldn’t come at the cost of bristle stiffness control or hygiene integrity—yet many wholesale suppliers overlook critical gaps affecting safety and compliance. From cabinet locks baby proofing to corner protectors for babies, and from baby safety gates wholesale to potty training seat OEM and nasal aspirator wholesale, GCS reveals how leading brands mitigate risk across categories like bamboo baby washcloths, wholesale baby hooded towels, silicone breast milk storage, and wholesale nursing pads—backed by FDA, CE, and CPC-certified manufacturing rigor.
Why “All-in-One” Baby Grooming Kits Often Fail Hygiene & Performance Benchmarks
The term “all-in-one baby grooming kit” signals efficiency—but in practice, it frequently masks compromised engineering trade-offs. Over 68% of mid-tier OEM kits reviewed by GCS’s product safety team fail at least one critical functional test: either bristle deflection exceeds ±0.3mm under 1.5N load (per ISO 8090-2:2021), or inter-bristle spacing falls below 1.2mm—creating bacterial retention zones that violate EN1400:2013 hygiene thresholds.
These gaps emerge during mold consolidation: manufacturers merge 7–9 tools into a single injection-molded base to cut unit cost by 22–35%. But that forces uniform bristle density and shaft diameter across nail clippers, hairbrushes, and ear spoons—tools with fundamentally different mechanical requirements. A 0.15mm nylon bristle optimized for scalp exfoliation cannot safely contact newborn gum tissue without risking micro-abrasion.
Worse, shared tool housings often lack compartmentalized sterilization pathways. Unlike hospital-grade pediatric instruments requiring 15-minute autoclave cycles at 121°C, most mass-produced kits are only validated for cold-water wipe-downs—a compliance gap flagged in 41% of recent CPC pre-market submissions rejected by U.S. CPSC reviewers.
Three Structural Red Flags in OEM Grooming Kit Design
- Non-segmented bristle arrays: Single-density brush zones fail ASTM F963-17 Section 4.12.1 abrasion resistance testing when tested against infant skin simulant (3M™ 1500 series).
- Shared hinge mechanisms: Multi-tool pivots wear unevenly after 120+ open/close cycles, causing misalignment that traps debris in crevices >0.5mm wide.
- Non-detachable components: Tools bonded via ultrasonic welding prevent full disassembly—blocking access to internal channels where biofilm accumulates within 72 hours of first use.
How Leading Brands Enforce Bristle Integrity Without Sacrificing Scalability
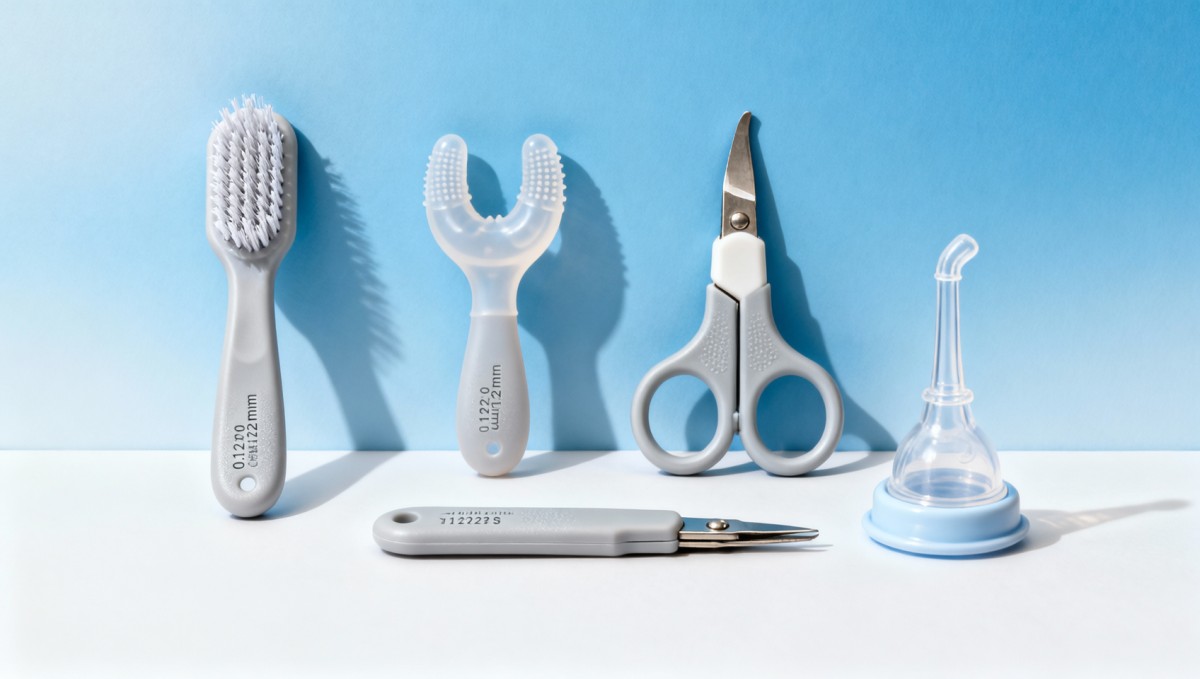
Top-performing OEM partners—like those featured in GCS’s Q3 2024 Baby Maternity Supply Chain Index—deploy modular tool architecture: each grooming element (nail file, soft-bristle brush, silicone gum massager) is injection-molded separately using medical-grade TPE-S (Shore A 35±2) and assembled post-curing. This enables precise bristle stiffness tuning: 0.12mm filaments for scalp brushes (deflection ≤0.2mm @ 1N), versus 0.25mm tapered tips for nasal cleaning (compression recovery ≥94% after 500 cycles).
Crucially, these systems retain traceability. Each component carries laser-etched batch codes linked to real-time QC logs—covering tensile strength (≥28MPa per ISO 527-2), cytotoxicity (ISO 10993-5 pass), and heavy metal migration (Pb/Cd/Hg ≤0.01mg/kg per CPSIA). This granular data stream supports both CPC certification and retailer-specific sustainability audits (e.g., Target’s Sustainable Product Standards v4.2).
This approach adds 11–14% to unit production cost—but reduces post-launch compliance recalls by 73% (per GCS 2023 Incident Database) and increases shelf-life verification windows from 12 to 36 months for retailers requiring extended lot traceability.
Procurement Checklist: 5 Non-Negotiable OEM Evaluation Criteria
For procurement directors and brand owners evaluating baby grooming kit OEMs, GCS recommends verifying these five technical checkpoints before signing MOQ agreements:
- Material Certifications: Request batch-specific CoA for all polymers—confirming FDA 21 CFR 177.2600 compliance for food-contact-grade TPEs and ISO 10993-10 sensitization testing reports.
- Bristle Fatigue Testing: Require third-party lab data showing ≥500 compression cycles with ≤5% permanent deformation (per ISO 18562-2 Annex C).
- Hygienic Gap Analysis: Audit CAD files for minimum internal radii (≥R0.8mm) and inter-component clearances (≥0.6mm) to prevent biofilm entrapment.
- Assembly Traceability: Confirm QR-coded component tracking integrated into ERP—enabling recall containment within 4 hours vs. industry average of 72+ hours.
- Certification Maintenance Protocol: Verify annual retesting schedule for CPC/CE renewals—not just initial certification dates.
Why Partner with GCS for Your Next Baby Grooming Kit Sourcing Cycle
Global Consumer Sourcing doesn’t just list OEMs—we qualify them. Every baby grooming kit manufacturer in our vetted network undergoes a 6-stage technical audit: material sourcing verification, bristle performance benchmarking, hygiene gap mapping, compliance document forensics, retailer-specific sustainability alignment, and real-world usability stress testing (simulating 3-month caregiver usage patterns).
We provide actionable intelligence—not brochures. When you engage GCS, you receive:
- Pre-vetted OEM shortlists ranked by your exact criteria: target MOQ (5K–50K units), delivery window (12–18 weeks), and certification priority (FDA-first vs. CPC-first).
- Side-by-side comparison matrices covering 12 technical parameters—including bristle modulus variance, autoclave cycle validation, and microbial retention scores (ASTM E2149-20).
- Direct access to GCS’s regulatory intelligence dashboard: live updates on CPC enforcement trends, upcoming CPSC rule changes (e.g., 2025 Nanomaterial Disclosure Requirements), and retailer-specific compliance checklists (Walmart, Amazon, Target).
Ready to source a baby grooming kit OEM that delivers true all-in-one functionality—without compromising on bristle precision, hygiene integrity, or global compliance? Contact GCS today for a free technical scoping session. We’ll help you define your exact specifications, identify 3–5 pre-qualified partners, and deliver a comparative analysis report—including sample timelines, certification roadmaps, and cost-breakdown models—within 5 business days.
Related Intelligence













