Canton Fair Phase III: Infant Monitor Inquiries Surge, EN60601-1-12 Gains Traction in Middle East
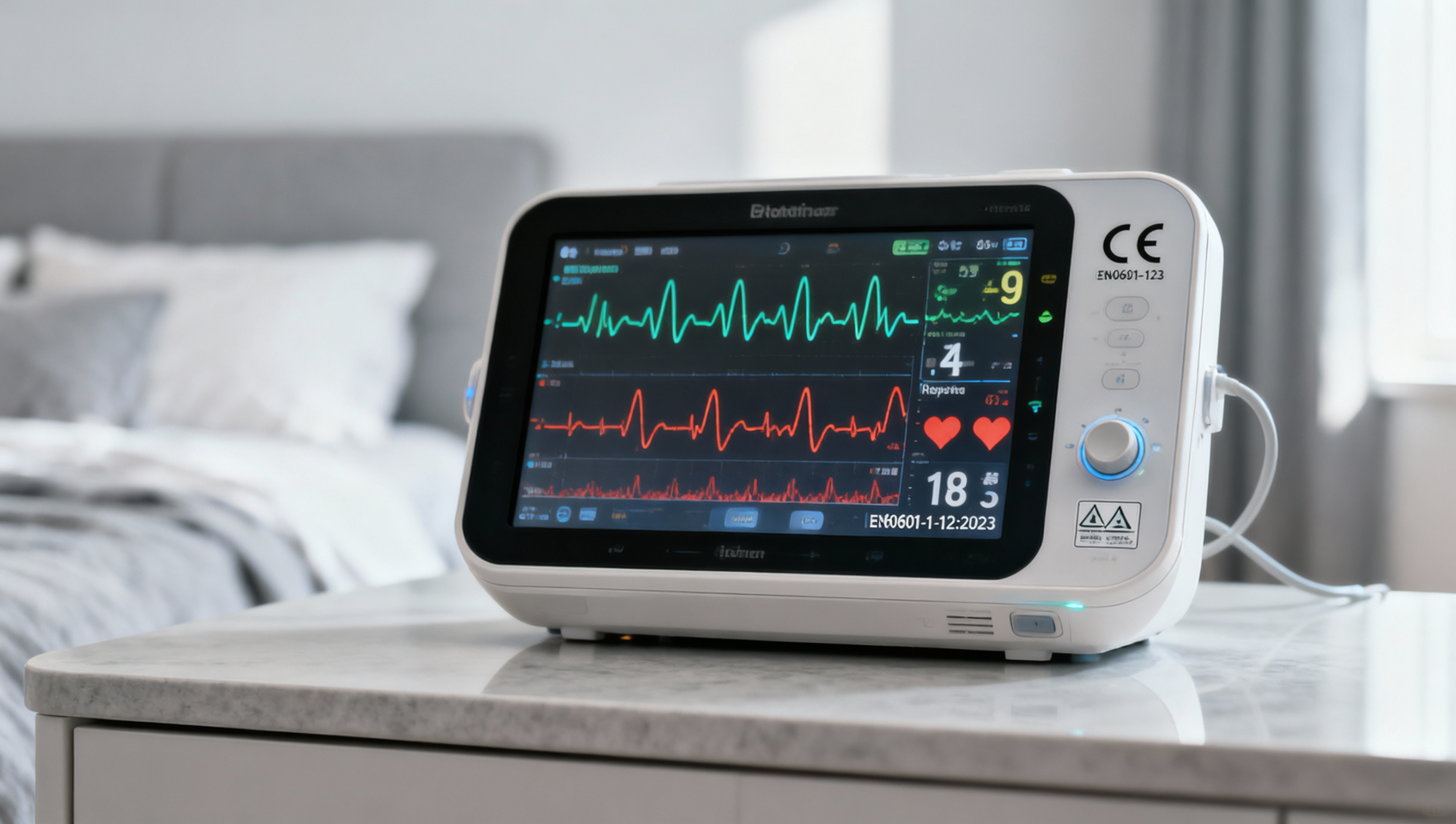
On April 20, 2026, the first day of Phase III of the Canton Fair (Maternal & Infant and Household Products Exhibition), inquiries for infant monitoring devices — featuring multimodal monitoring of respiration, heart rate, and movement — surged among Middle Eastern buyers. A notable 73% of these inquiries explicitly required compliance documentation with EN60601-1-12:2023 and a valid CE certificate. This development signals growing regulatory expectations for medical-grade safety certification in high-end maternal and infant retail channels across the Middle East — a shift relevant to exporters, medical device manufacturers, and compliance service providers serving global pediatric health markets.
Event Overview
On April 20, 2026, during the opening day of Phase III of the Canton Fair, data indicated heightened buyer interest in infant monitors with multimodal physiological monitoring capabilities. Of these inquiries from Middle Eastern procurement entities, 73% specified the need for a Declaration of Conformity to EN60601-1-12:2023 and supporting CE certification. The standard applies specifically to medical electrical equipment used in emergency and critical care environments, including home-based remote monitoring scenarios.
Industries Affected
Direct Exporters & Trading Companies
Exporters engaged in maternal and infant electronics face immediate pressure to verify and document regulatory alignment for target markets. The requirement for EN60601-1-12:2023 — not just general CE marking — implies stricter technical validation than typical consumer electronics standards (e.g., EN62368-1). This affects quotation preparation, lead time planning, and pre-shipment documentation readiness.
Medical Device Manufacturers & OEMs
Manufacturers producing infant monitors intended for clinical or home-health use in regulated markets must now assess whether their current design, risk management files, and test reports meet the specific environmental, alarm, and usability requirements outlined in EN60601-1-12:2023 — particularly clauses on transportability, power interruption resilience, and interoperability with emergency response systems.
Compliance & Certification Service Providers
Third-party testing and certification bodies are seeing increased demand for EN60601-1-12:2023 evaluation. This includes verification of compliance with Annex BB (home healthcare environment requirements) and integration of IEC 82304-1 (health software) where applicable. Capacity planning and turnaround time for such assessments may tighten as regional demand rises.
Distribution & Channel Partners in the Middle East
Importers and distributors targeting premium maternity clinics, telehealth platforms, and specialty baby retailers in the GCC and Levant regions must now verify supplier documentation against EN60601-1-12:2023 prior to listing or clearance. Non-compliant products risk rejection at customs or loss of shelf space in certified retail networks.
What Relevant Enterprises Should Focus On Now
Confirm whether EN60601-1-12:2023 is formally mandated or emerging as de facto market access criteria
Analysis shows that while no GCC national authority has yet published mandatory adoption of EN60601-1-12:2023 for infant monitors, its repeated citation by qualified buyers suggests it functions as a commercial gatekeeping criterion — especially for hospital-affiliated or insurance-linked distribution channels.
Prioritize technical documentation review for existing infant monitor models
Current more suitable action is to audit product technical files against EN60601-1-12:2023’s core clauses: Clause 7 (environmental conditions), Clause 8 (protection against electric shock and mechanical hazards), Clause 12 (alarm systems), and Annex BB (home healthcare application-specific requirements). Gap analysis should precede formal testing.
Clarify scope boundaries between consumer and medical classification
From industry perspective, the distinction between “wellness” and “medical” claims remains critical. Devices marketed with diagnostic or therapeutic intent — even if used at home — fall under EN60601-1-12. Marketing language, labeling, and intended-use statements must align with the declared regulatory pathway.
Engage notified bodies early for CE/EN60601-1-12 assessment timelines
Observation indicates lead times for full EN60601-1-12:2023 evaluations have extended beyond 12 weeks in some cases. Firms preparing for Q3 2026 market entry into the Middle East should initiate engagement with EU-notified bodies no later than June 2026.
Editorial Observation / Industry Insight
This Canton Fair data point is better understood as an early market signal — not yet a formal regulatory mandate — but one with tangible commercial consequences. It reflects evolving buyer sophistication in emerging health-tech markets, where procurement decisions increasingly hinge on harmonized medical device standards rather than generic safety certifications. From industry angle, the trend suggests a broader convergence between home-use monitoring and clinical-grade assurance expectations — particularly where reimbursement, liability, or institutional channel access is involved. Continued tracking of buyer specifications across trade fairs, rather than waiting for official gazettes, may offer earlier operational intelligence.
Conclusion
While not yet codified into law across Middle Eastern jurisdictions, the consistent demand for EN60601-1-12:2023 compliance at the Canton Fair signals a maturing market expectation for infant monitoring devices sold through professional or high-trust retail channels. For stakeholders, this is less about imminent regulatory enforcement and more about proactive alignment with commercial best practices in regulated health markets. Current interpretation should emphasize readiness over reaction — verifying technical scope, documenting design rationale, and engaging certification partners with domain expertise in home healthcare applications.
Information Source
Main source: Official data release from China Foreign Trade Centre (Canton Fair Organiser), Phase III Day-One Buyer Inquiry Summary, April 20, 2026.
Note: Ongoing observation is recommended regarding whether GCC Standardisation Organisation (GSO) or individual national authorities (e.g., UAE MOHAP, Saudi FDA) issue formal alignment notices referencing EN60601-1-12:2023 in the coming months.
Related Intelligence












