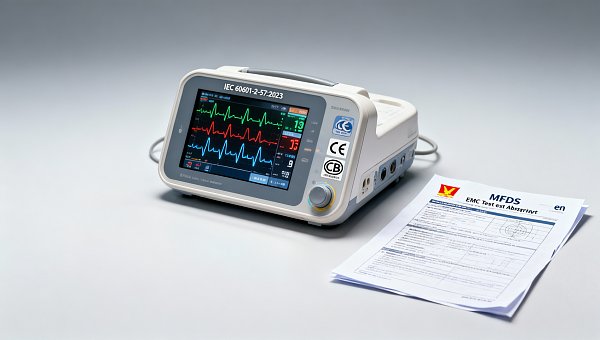
Vietnam MFDS Temporarily Waives EMC Pre-Cert for Smart Monitors
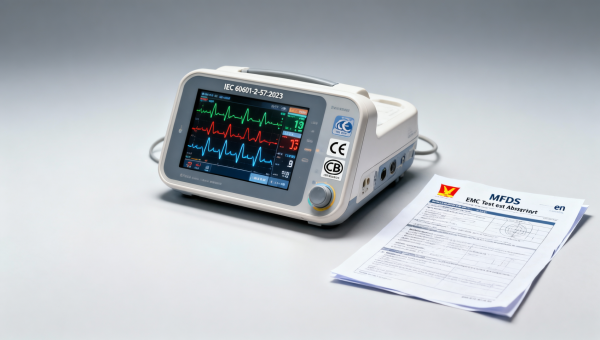
On May 12, 2026, Vietnam’s Ministry of Food and Drug Safety (MFDS) issued an emergency notice temporarily waiving the electromagnetic compatibility (EMC) pre-certification requirement for smart infant and elderly monitors compliant with IEC 60601-2-57:2023. This regulatory adjustment is expected to reduce export lead times by approximately 14 days for Chinese manufacturers supplying such devices to Vietnam — a development particularly relevant to medical device exporters, EMC testing service providers, and regulatory compliance teams operating in ASEAN market access.
Event Overview
On May 12, 2026, Vietnam’s Ministry of Food and Drug Safety (MFDS) published an emergency notice stating that, for smart infant/elderly monitors conforming to IEC 60601-2-57:2023, the EMC pre-certification step is temporarily waived. Under this measure, applicants may register these devices using a valid CB test report plus a local Vietnamese EMC test summary — without submitting full pre-certified EMC documentation. The notice is effective immediately and applies only to the specified product category and standard.
Industries Affected
Medical Device Exporters (Direct Trade Enterprises)
These enterprises are directly affected because the waiver modifies a mandatory registration prerequisite. The impact manifests primarily in shortened time-to-market: reduced administrative review cycles and faster clearance at Vietnam’s regulatory gateway. For exporters managing multiple ASEAN submissions, this change may enable more predictable shipment scheduling and improved responsiveness to tender deadlines.
EMC Testing & Regulatory Compliance Service Providers
Service providers supporting Vietnamese market entry face a shift in scope and deliverables. While full pre-certification is waived, local EMC test summaries remain required — meaning labs must still perform targeted EMC measurements in Vietnam or via authorized local partners. The impact lies in altered service bundling (e.g., less emphasis on full EMC certification packages, more focus on summary report generation and local lab coordination).
OEM/ODM Manufacturers of Smart Health Monitors
Manufacturers producing under contract for global brands are impacted operationally: they must verify whether their current IEC 60601-2-57:2023-compliant designs meet the exact scope defined in the MFDS notice (e.g., function, classification, software architecture). Any deviation — such as inclusion of wireless charging or non-standard connectivity modules — may disqualify the device from the waiver, requiring full EMC pre-certification regardless.
Key Considerations and Recommended Actions
Monitor official updates for scope clarification or extension
The notice is labeled “emergency” and does not specify an end date. Companies should track subsequent MFDS communications for possible expansion to other IEC 60601-2-x standards, or for formalization into a permanent guideline. Subscribing to MFDS’s official notification channel and engaging local regulatory agents is advisable.
Verify device eligibility against the exact standard and use case
The waiver applies exclusively to devices complying with IEC 60601-2-57:2023 and explicitly designated for infant or elderly monitoring. Devices marketed for clinical diagnostics, therapeutic support, or general wellness — even if technically similar — fall outside the exemption. Companies should conduct internal eligibility reviews before initiating registration.
Distinguish between policy signal and operational readiness
While the waiver removes one procedural step, other registration requirements (e.g., technical file submission, local representative appointment, labeling compliance) remain unchanged. Teams should avoid assuming overall simplification; instead, treat the EMC waiver as a discrete efficiency gain within an otherwise stable framework.
Prepare documentation workflows for local EMC test summaries
Since third-party CB reports alone are insufficient, manufacturers must secure local EMC test summaries from Vietnam-accredited labs. This requires advance coordination — including sample dispatch, test planning, and translation/localization of summary reports. Integrating this step into existing pre-submission checklists helps prevent bottlenecks.
Editorial Perspective / Industry Observation
Observably, this measure reflects Vietnam’s growing emphasis on streamlining market access for low-risk, standards-aligned health technologies — particularly those addressing demographic priorities like aging populations and infant care. Analysis shows it is best understood not as a broad deregulatory shift, but as a targeted, time-bound facilitation tool. From an industry perspective, its significance lies less in precedent-setting reform and more in immediate supply chain responsiveness: it lowers friction for a specific, high-volume export segment without altering core safety expectations. Continued attention is warranted, however, as similar waivers could emerge for adjacent categories (e.g., wearable vital sign trackers) if this trial proves administratively sustainable.
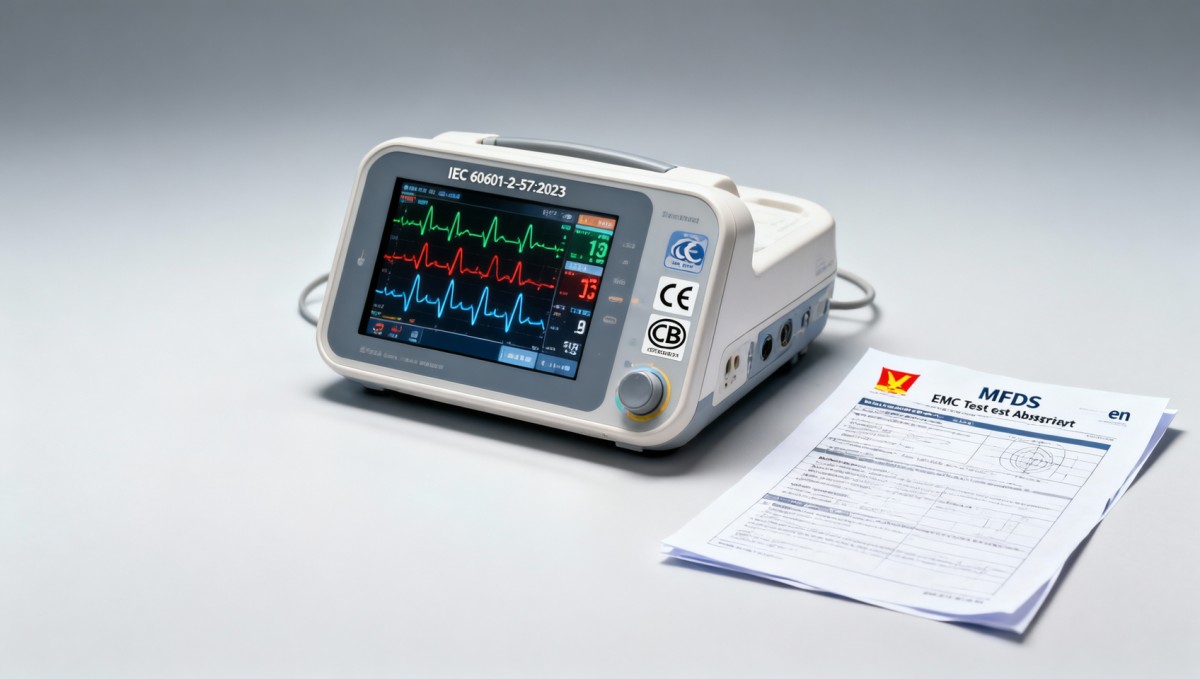
Conclusion
This notice represents a concrete, short-term improvement in regulatory efficiency for a defined subset of smart health monitoring devices entering Vietnam. Its value is operational rather than structural: it accelerates timelines without relaxing technical or clinical safety requirements. Currently, it is more accurately interpreted as a tactical adjustment to administrative process — not a signal of broader regulatory liberalization. Stakeholders are advised to apply it precisely as written, maintain vigilance on its duration and scope, and avoid extrapolating its implications beyond the IEC 60601-2-57:2023-based infant/elderly monitor category.
Source: Official emergency notice published by Vietnam Ministry of Food and Drug Safety (MFDS), dated May 12, 2026.
Note: The notice’s expiration date, potential renewal, or expansion to other standards remains unconfirmed and is subject to ongoing observation.
Related Intelligence