RCEP Members Adopt ASEAN-FFL 2026 for Infant Feeding Products
On 27 April 2026, RCEP member economies jointly issued the ASEAN Unified Labelling Guide for Infant Feeding Products (ASEAN-FFL 2026), introducing mandatory front-of-pack labelling requirements for silicone pacifiers, temperature-controlled baby bottles, and infant food grinders. The regulation directly affects manufacturers, exporters, and distributors operating across Vietnam, Thailand, and Indonesia — markets where enforcement begins 1 December 2026. This development signals a coordinated shift toward harmonized safety transparency in infant product trade across the RCEP-ASEAN interface.
Event Overview
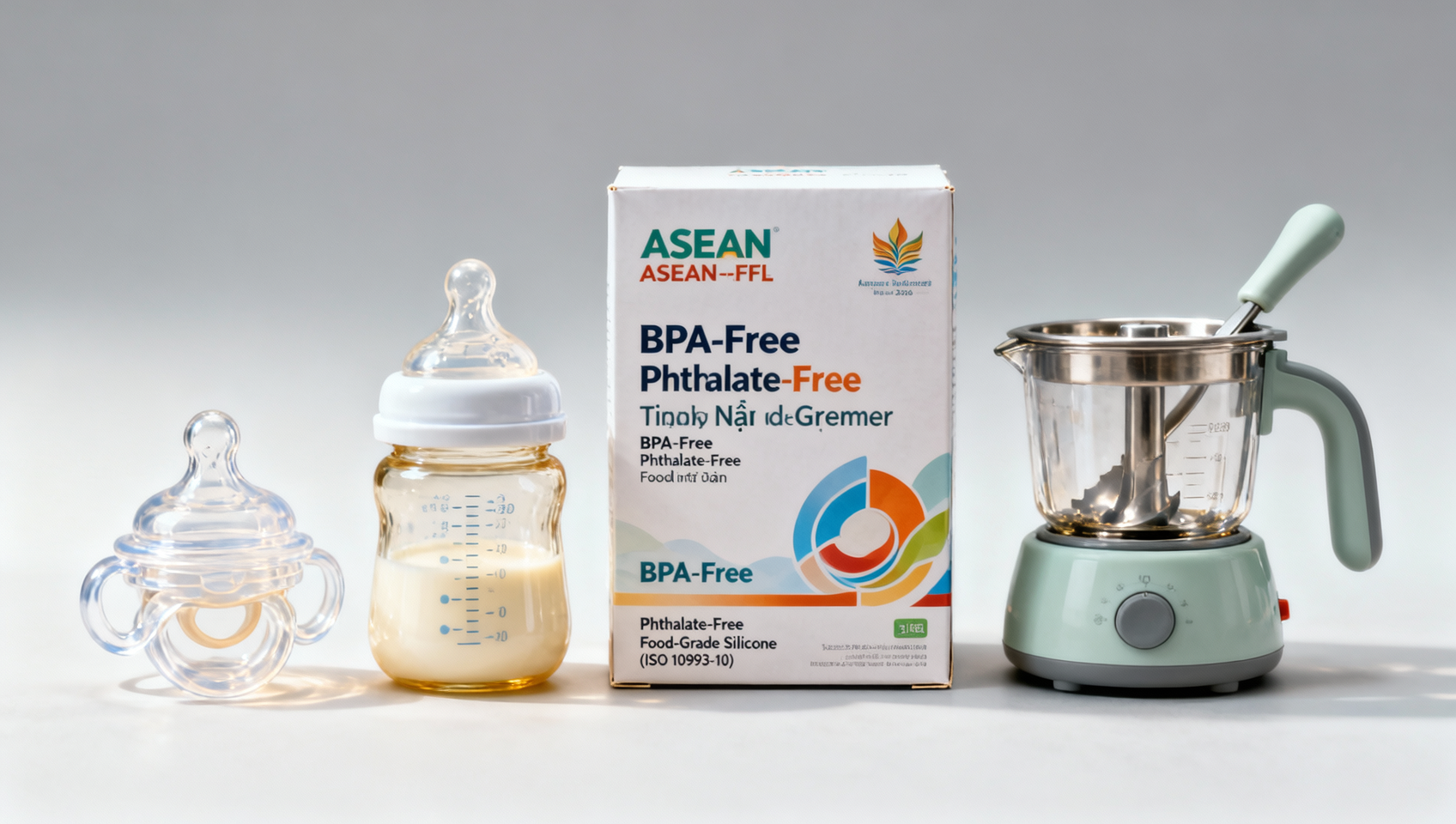
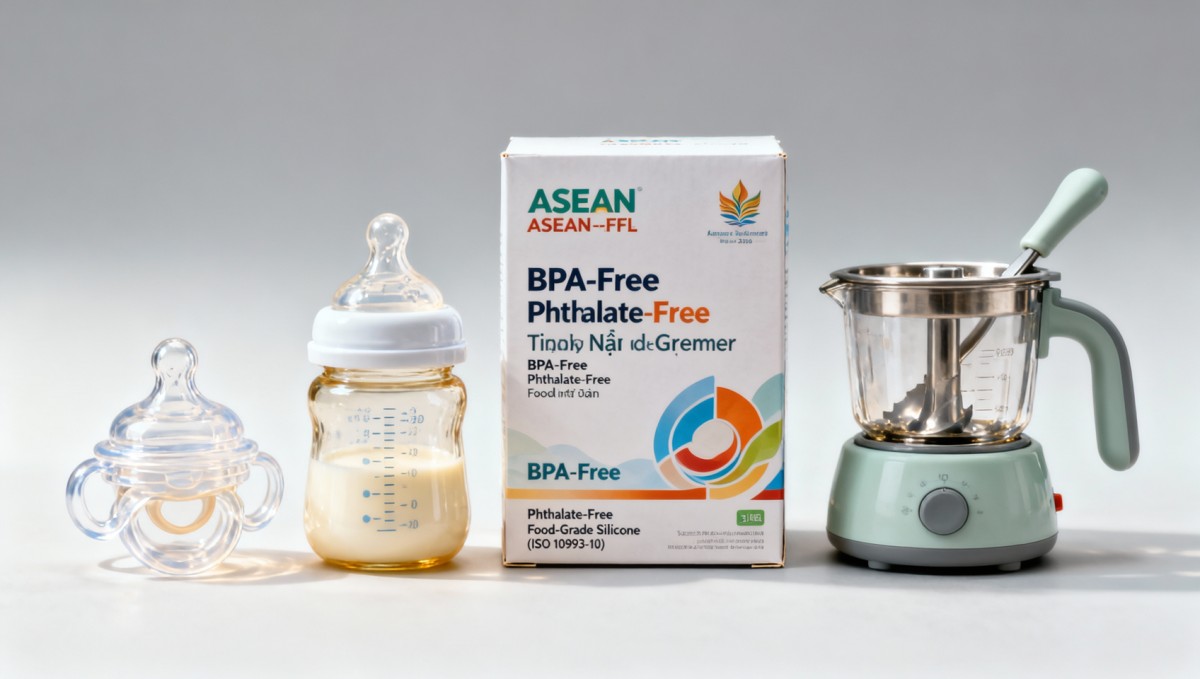
On 27 April 2026, the RCEP Secretariat and the ASEAN Advisory Committee on Standards and Quality (ASEAN-ACSQ) jointly published the ASEAN Unified Labelling Guide for Infant Feeding Products (ASEAN-FFL 2026). The guide mandates that products including silicone pacifiers, temperature-controlled baby bottles, and infant food grinders must display, on the primary packaging face, three statements in font size no smaller than 12 pt: ‘BPA-Free’, ‘Phthalate-Free’, and ‘Food-Grade Silicone (ISO 10993-10)’. These statements must appear simultaneously in English, Chinese, Vietnamese, Thai, and Indonesian. The regulation enters into force on 1 December 2026 for Vietnam, Thailand, and Indonesia.
Which Subsectors Are Affected
Direct Exporters & Importers of Infant Feeding Products
These businesses face immediate compliance obligations when shipping to Vietnam, Thailand, or Indonesia. Labelling deviations may result in customs rejection, rework delays, or market access suspension. Impact is concentrated at the final packaging and documentation stage — particularly for multi-market SKUs where one label design previously served several jurisdictions.
Raw Material Suppliers (e.g., Silicone Compound Producers)
Suppliers must now provide certified documentation confirming ISO 10993-10 biocompatibility for food-contact silicone grades supplied to infant product assemblers. Absence of test reports aligned with ASEAN-FFL 2026’s cited standard may interrupt downstream production or trigger audit requests from brand owners.
OEM/ODM Manufacturers & Contract Assemblers
Manufacturers producing under private labels or white-label agreements must revise artwork templates, update packaging line settings (e.g., print font sizing), and validate multilingual label layouts prior to 1 December 2026. No grandfathering is indicated for existing stock; newly shipped units must comply regardless of production date.
Distribution & E-commerce Platforms Serving ASEAN Markets
Platforms listing infant feeding products for sale in Vietnam, Thailand, or Indonesia will need to verify label compliance before enabling checkout. Non-compliant listings risk takedown or penalties under national consumer protection frameworks now referencing ASEAN-FFL 2026 as a benchmark.
What Relevant Businesses or Practitioners Should Focus On Now
Monitor official implementation guidance from national standards bodies
While ASEAN-FFL 2026 is published, national adoption mechanisms — such as Vietnam’s Ministry of Health or Thailand’s FDA — have not yet released detailed enforcement protocols or transitional arrangements. Businesses should track announcements from these authorities, especially regarding grace periods, verification procedures, and acceptable evidence formats for ‘BPA-Free’ and ‘Phthalate-Free’ claims.
Prioritise compliance review for high-volume export SKUs targeting Vietnam, Thailand, and Indonesia
Not all infant feeding products are equally impacted. Focus first on silicone pacifiers, temperature-controlled bottles, and manual food grinders — the only categories explicitly named in the guide. Review current label artwork, font specifications, language coverage, and certification alignment against ISO 10993-10 for those SKUs.
Distinguish between regulatory signal and enforceable requirement
ASEAN-FFL 2026 is a harmonised guideline adopted by RCEP members, not an ASEAN treaty-level standard. Its legal weight derives from national transposition — meaning its enforceability depends on domestic legislation. Until local laws reference ASEAN-FFL 2026 explicitly, it functions primarily as a strong policy signal and de facto expectation.
Prepare updated packaging assets and supplier declarations ahead of Q3 2026
Reprinting packaging requires lead time — typically 6–8 weeks for multilingual flexographic plates or digital label runs. Concurrently, request updated material safety data sheets and ISO 10993-10 test certificates from silicone and plastic component suppliers. Internal cross-functional alignment (regulatory affairs, procurement, QA, logistics) should begin immediately.
Editorial Perspective / Industry Observation
Observably, ASEAN-FFL 2026 reflects a broader regional trend: the consolidation of chemical safety disclosures into visible, consumer-facing claims — moving beyond technical documentation to front-of-pack clarity. Analysis shows this is less about introducing new chemical restrictions (BPA and phthalates are already banned or limited in most RCEP markets) and more about standardising how compliance is communicated across borders. From an industry perspective, it functions primarily as a coordination mechanism — reducing label fragmentation but increasing upfront verification burden. It is currently best understood as a binding operational signal rather than a fully implemented legal regime, given its dependence on national adoption timelines and enforcement capacity.
Conclusion
ASEAN-FFL 2026 marks a step toward regulatory convergence in infant product labelling across key RCEP-ASEAN markets, but its practical impact remains contingent on national implementation. For industry stakeholders, it is more accurately interpreted as a near-term operational checkpoint — requiring targeted label and documentation updates — rather than a fundamental shift in material safety requirements. A measured, market-specific, and documentation-first response aligns best with the current state of the guideline.
Information Sources
— RCEP Secretariat Official Notice (27 April 2026)
— ASEAN Advisory Committee on Standards and Quality (ASEAN-ACSQ), ASEAN-FFL 2026 Guide
Note: National transposition timelines, enforcement details, and potential extensions for small enterprises remain under observation and are not yet publicly confirmed.
Related Intelligence