Canton Fair Phase III: Baby Monitors Surge, EN60601-1-12 Focus in Middle East
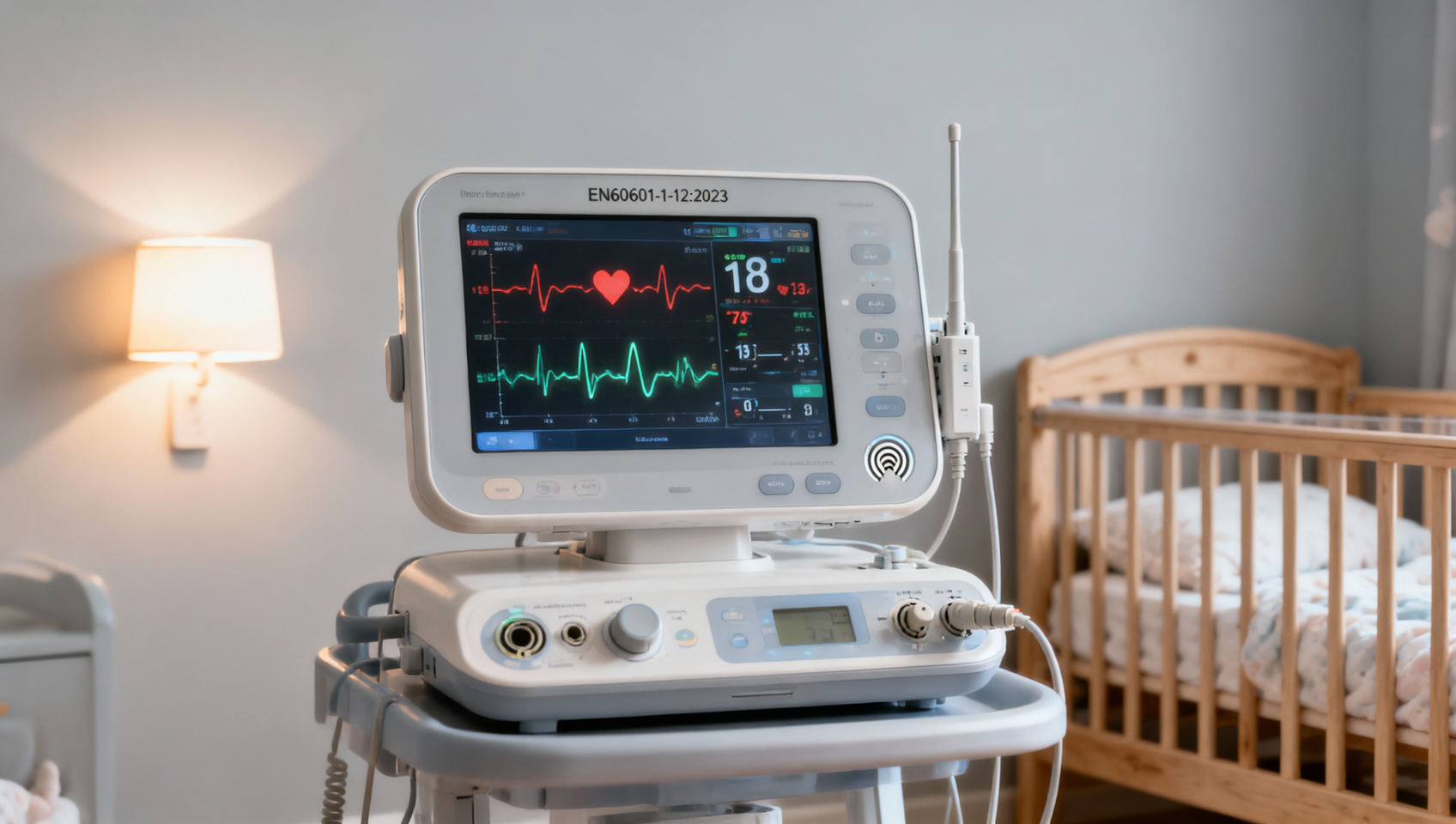
On April 22, 2026, the opening day of Phase III of the Canton Fair (Maternal & Infant Care and Home Products Exhibition), inquiry volume for baby monitors rose 142% year-on-year. Middle Eastern buyers showed concentrated interest in compliance with EN60601-1-12:2023 — the latest medical electrical equipment safety standard covering remote monitoring and wireless communication requirements. This development signals heightened regulatory awareness among export-oriented nursery furniture and monitor manufacturers in China.
Event Overview
On April 22, 2026, the third phase of the Canton Fair commenced, focusing on maternal, infant, and home products. According to on-site reporting from the exhibition, inquiries for baby monitors increased by 142% compared to the same period in the previous year. A notable pattern emerged: procurement delegations from the Middle East prioritized verification of conformity with EN60601-1-12:2023, particularly its provisions on remote monitoring functionality and wireless communication safety. No further quantitative or qualitative data beyond this has been officially confirmed.
Industries Affected
Export-Oriented Baby Monitor Manufacturers
These firms face direct pressure to align product design, testing, and documentation with EN60601-1-12:2023. The surge in targeted inquiries indicates that non-compliant units may encounter reduced market access or extended negotiation cycles in regulated Middle Eastern markets.
Nursery Furniture Producers Integrating Monitoring Features
Manufacturers embedding sensors or connectivity modules into cribs, bassinets, or nursery furniture are now subject to medical-grade safety expectations — not just consumer electronics standards. Their certification pathways may require re-evaluation of risk management files and electromagnetic compatibility (EMC) test scope.
Third-Party Testing and Certification Service Providers
Demand for EN60601-1-12:2023 validation is rising among Chinese exporters. Labs accredited for IEC/EN60601 series standards — especially those with recognized status under EU Notified Body frameworks — may see increased workload and tighter scheduling for pre-market assessments.
Distributors and Brand Owners Targeting GCC Markets
Importers and regional brand holders must now verify supplier documentation against EN60601-1-12:2023 before customs clearance or shelf placement in Gulf Cooperation Council (GCC) countries. Failure to confirm compliance may delay approvals under local medical device regulations (e.g., SFDA in Saudi Arabia or MOH in UAE).
What Relevant Enterprises Should Focus On Now
Monitor official updates from GCC national regulators
While EN60601-1-12:2023 is referenced by buyers, formal adoption as a mandatory requirement varies across GCC jurisdictions. Companies should track published notices from Saudi FDA, UAE MOHAP, and Qatar’s MOPH regarding classification thresholds for ‘medical-grade’ infant monitors.
Verify whether your product falls under medical device scope in target markets
Not all baby monitors are automatically classified as medical devices. Classification depends on intended use claims (e.g., ‘apnea detection’, ‘heart rate trending’) and technical capabilities. Firms should conduct a formal regulatory determination before initiating certification.
Review existing test reports for alignment with EN60601-1-12:2023 clauses
Many manufacturers hold older EN60601-1:2012 or IEC60601-1:2012 certifications. These do not cover the 2023 edition’s new requirements for remote monitoring systems, cybersecurity considerations, and wireless coexistence. Gap analysis is required before renewal or extension.
Prepare technical documentation for potential conformity assessment
EN60601-1-12:2023 requires comprehensive risk management (ISO 14971), usability engineering (IEC 62366-1), and clinical evaluation documentation — even for low-risk applications. Exporters should begin assembling these elements ahead of formal application.
Editorial Observation / Industry Insight
From industry perspective, this trend reflects a broader shift: infant wellness technology is increasingly converging with medical device expectations in high-regulation markets. The Canton Fair data point does not yet indicate formal regulatory enforcement — but rather early-stage buyer due diligence behavior. Analysis来看, it functions less as an immediate compliance deadline and more as a leading indicator of tightening market gatekeeping. Observation来看, the focus on EN60601-1-12:2023 suggests buyers are proactively filtering suppliers based on readiness for future regulatory alignment — not just current certification status. Current interpretation is better understood as a signal of evolving procurement criteria, not a finalized policy change.
This is not yet a de facto standard requirement across the region, but one gaining traction among informed purchasers. Continued observation is warranted over the next 6–12 months to assess whether such inquiries translate into contractual obligations or national-level regulatory updates.
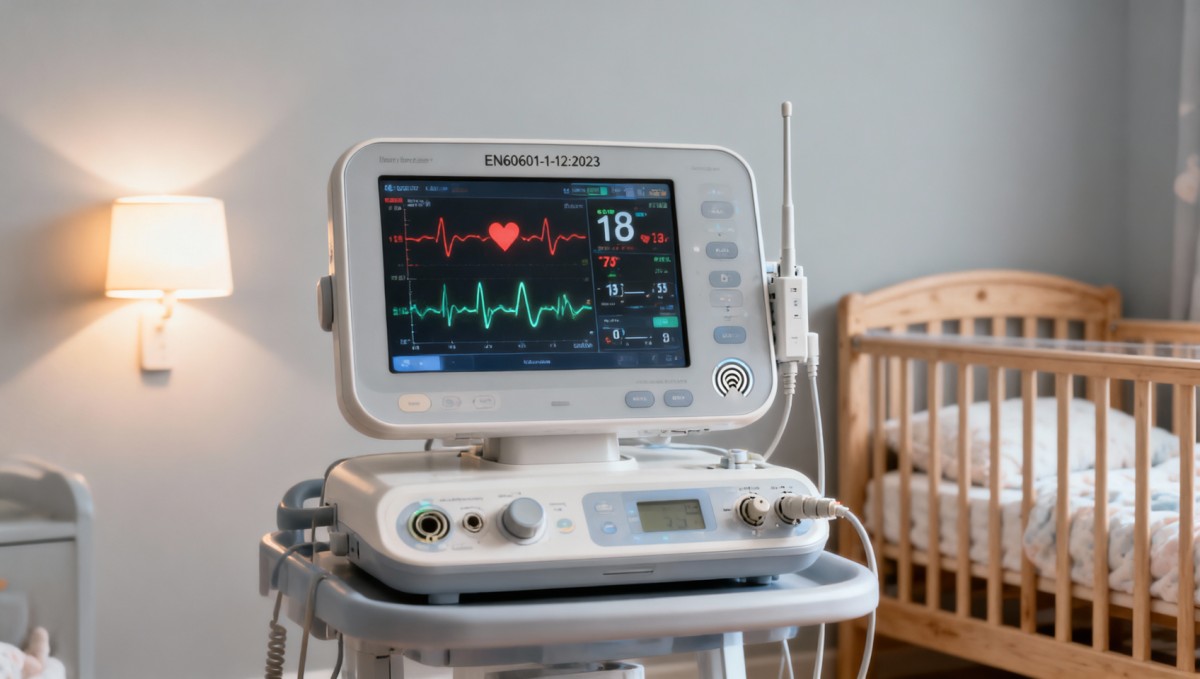
In summary, the April 22, 2026 Canton Fair data highlights a measurable uptick in demand-side scrutiny around medical-grade safety for baby monitors — particularly in the Middle East. It underscores how trade fairs serve as real-time barometers of regulatory anticipation, not just sales platforms. For stakeholders, the value lies not in reacting to a new rule, but in recognizing an emerging expectation — and adjusting technical preparation accordingly.
Source: Official Canton Fair daily briefing (Phase III, April 22, 2026); On-site buyer interviews reported by fair organizers; Publicly available EN60601-1-12:2023 standard text (CENELEC).
Note: GCC national implementation timelines and enforcement mechanisms remain unconfirmed and are subject to ongoing observation.
Related Intelligence












